12 Dec 2016
Paul Horwood discusses some of the cattle products available on the market and methods on how to prevent a disease outbreak.

Figure 1. Bovine viral diarrhoea remains a very common disease in the UK.
Decision making on vaccination protocols for UK farms can be complicated, with an array of different products available, and each client having different diseases present, management decisions regarding biosecurity and attitudes to risk.

With the squeeze on farm gate prices, farmers are looking to justify every pound spent and medicine spend is under the spotlight. The old adage of prevention being better than cure obviously holds true, but it is important to regularly review the vaccine protocols.
Changes in biosecurity, or a drive to eradicate certain diseases, can mean vaccine use could be reduced; alternatively, a drive to increase herd size and buying in of cattle could result in a need to increase vaccination.
Bovine viral diarrhoea (BVD) remains a very common disease in the UK, despite vaccines being available for more than 15 years. About 60% of cattle in the EU test positive for exposure to the BVD virus (Lindberg et al, 2006) and, although BVD type-one is the most common form, outbreaks of BVD type-two were confirmed in 2013 and 2014 in Belgium, Germany and the Netherlands.
BVD type-two is generally a more serious disease with high levels of mortality and, while the virus has been identified in the UK, it has not yet been recognised as the cause of serious outbreaks of disease in cattle.
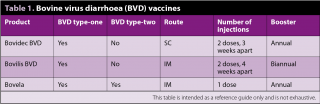
Some of the failures associated with BVD vaccination have been due to incorrect timing or failure to follow the two-shot programme correctly. The advent of a single-shot primary course being launched into the UK will protect against both types of BVD (Table 1).
Timings for all BVD vaccines are to aim for full protection to occur at least two to three weeks prior to service, to ensure full fetal protection and eliminate the risk of the heifer producing a persistently infected fetus.
Infectious bovine rhinotracheitis is a herpesvirus and, once infected, the animal remains infected for life, with the potential to spread the virus again during times of stress or immune suppression.
The advent of marker vaccines, which allows for serological testing to differentiate vaccinated from wild-type infected animals, has allowed for better screening of animals.
Live and inactivated vaccines are available. Live vaccines have rapid onset of immunity and are of use in the face of an outbreak to reduce clinical signs and improve the immunity quickly. Inactivated vaccines appear to be better at producing longer-term immunity and seem to reduce shedding from latently infected animals.
It is usual to recommend the inactivated vaccines in herd eradication schemes.
Leptospirosis is spread most often during the spring and summer months while cattle are at pasture. Hardjo serovars of Leptospira species are not carried by wildlife, but sheep can carry and excrete these serovars; therefore, mixed grazing is a risk factor. Two vaccines are available in the UK for leptospirosis control. In both cases a, primary course of two doses four to six weeks apart, followed up with annual boosters, is preferably given in the spring before the period of highest risk.
A number of vaccines are available for immunising pregnant cows and heifers to raise antibodies to rotavirus, coronavirus and Escherichia coli. After birth, the calves gain protection in their gut from drinking the colostrum and milk that is fortified with these antibodies.
Some minor differences exist in the timing of the various vaccines.
One is a single dose given between 3 and 12 weeks pre-calving and needs to be repeated for every subsequent pregnancy.
Another is a two-dose programme, with the first injection at one to two months before calving and the second injection two to four weeks after first injection, at least two weeks prior to calving.
A third vaccine is also a two-dose programme given during the later stages of pregnancy, with an interval of four to five weeks between doses and allowing two to three weeks from the time of the second dose until the predicted date of calving. During each subsequent pregnancy, previously vaccinated cows should receive a single injection two to six weeks prior to the predicted calving date.
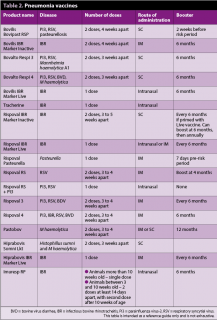
Calf pneumonia vaccines are available for infectious bovine rhinotracheitis, parainfluenza type-three, bovine respiratory syncitial virus, Pasteurella, Mannheimia haemolytica and Histophillus somni, and many combinations are available depending on what protection is needed.
It is important to identify the common diseases present and the age calves become infected (Table 2).
The only lungworm vaccine available uses irradiated live lungworm larvae that create an immune response from the animal, but the larvae do not continue to reproduce, so do not cause clinical disease.
The vaccine is a two-dose programme given approximately four weeks apart to youngstock at the beginning of their first grazing season. The second dose should be given at least two weeks before turnout, and vaccinated and unvaccinated stock should not be mixed for at least two weeks after the second dose has been given.
It is preferable for calves to be exposed to low levels of lungworm larvae throughout the grazing season to stimulate and maintain this immunity. If the worming protocol on the farm is too effective, there may be small exposure of the calves to lungworm, allowing very little natural immunity to build up, and this leads to insufficient long-term immunity in adult cattle, which can develop the disease in subsequent grazing years.
The vaccine should not be used in animals suspected of having clinical lungworm disease as this may make the clinical signs worse. These should be wormed and treated with antibiotics and anti-inflamatories, as necessary.
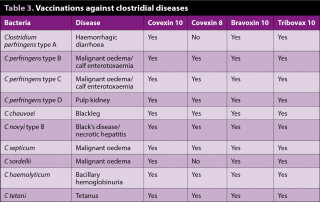
Vaccinations against clostridial diseases are routinely given to sheep, but the uptake is much less in the cattle sector. A number of products exist on the market and these require a two-shot programme, with a four to six-week gap between them (Table 3).
Ringworm in cattle is most commonly caused by Trichophyton verrucosum and a vaccine can be used to reduce clinical signs (prophylaxis) and shorten the recovery time (therapeutic). It can be given at different doses depending on the desired outcome (Table 4).
One mastitis vaccine can be used to reduce the incidence of subclinical mastitis, and the incidence and the severity of clinical mastitis caused by Staphylococcus aureus, coliforms and coagulase-negative staphylococci.
It is a three-dose programme, with the first injection at 45 days before the expected calving date, the second injection 35 days thereafter (about 10 days before the expected calving date) and the third injection 62 days after the second injection (about 52 days post-parturition).
The entire herd should be vaccinated, including in-calf heifers, and the aim is to improve immunity during the high-risk period from transition to peak lactation, and the full programme should be repeated for each subsequent lactation.
In the face of a Salmonella outbreak on farm, a fluid vaccine can be used to improve immunity to S enterica serovar Dublin and S enterica serovar Typhimurium.
For pregnant cows, this primary vaccination course can be given irrespective of the reproductive status. Any pregnant cows that have not calved within eight weeks of the second dose of vaccine should receive a further 5ml dose three to four weeks pre-calving.
Although the last outbreak in the UK was in 2007, it was estimated bluetongue would arrive in the UK this year. So far, this has not been identified.
Control can be aided by reducing the exposure of cattle to the Culicoides midge with fly control and housing, but the primary prevention strategy will be vaccination. Two vaccinations are available.
Firstly, the vaccine must have been produced, transported, delivered and stored in a way that ensures it is able to stimulate an appropriate immune response. At this year’s World Buiatrics Congress, a paper was presented that highlighted all 17 farms in the study had a fridge that recorded temperatures outside of 2°C and 80°C (Williams and Paixao, 2016).
Secondly, even if the vaccine is in a suitable condition when it is administered, the animal must be capable of mounting an adequate immune response, so care must be taken to avoid periods of stress, such as the day of arrival on a new farm, or other concurrent diseases that will alter the immune response and the future immunity of the animal.
Looking to the future, considerable interest exists in products that, although not vaccines, are able to stimulate the immune response, and reduce morbidity and mortality in flocks and herds.